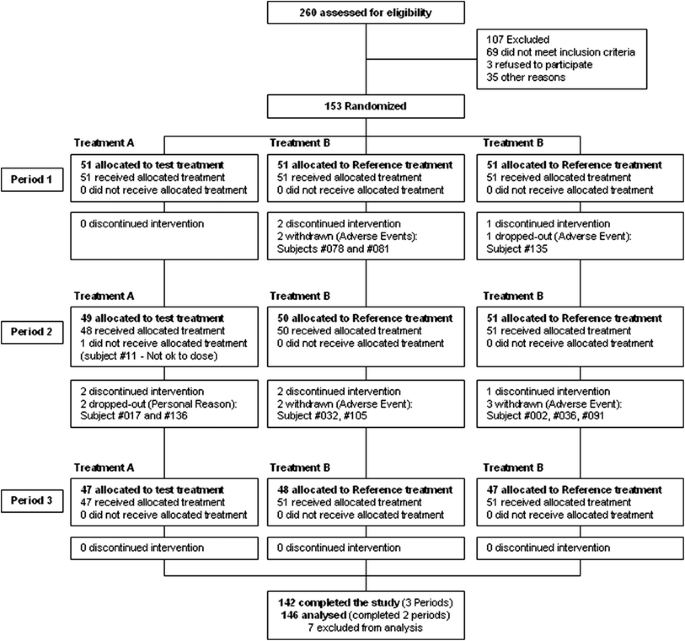
Bioequivalence Study of Two Formulations of Ibandronic Acid 150-mg Film-Coated Tablets in Healthy Volunteers Under Fasting Conditions: A Randomized, Open-Label, Three-Way, Reference-Replicated Crossover Study | SpringerLink
Sp.zn.sukls212402/2014 Příbalová informace: informace pro pacienta Ibandronic acid Teva 6 mg/6 ml koncentrát pro infuzní ro
ibandronic acid tablet | idrofos tablet 150 mg | ibandronic acid tablet uses in hindi | side effects - YouTube

PDF) Bioequivalence Study of Two Formulations of Ibandronic Acid 150-mg Film-Coated Tablets in Healthy Volunteers Under Fasting Conditions: A Randomized, Open-Label, Three-Way, Reference-Replicated Crossover Study
ИБАНДРОНИК АЦИД таблетки 150 мг * 1 ТЕВА (IBANDRONIK ACID tabl. 150 mg. * 1 TEVA), цена и информация
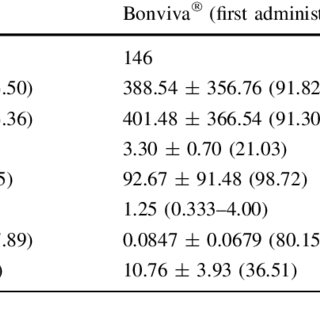







,aspect=fit)
,aspect=fit)

,aspect=fit)

